Can quantum simulations of the primordial soup finally crack the mother of all questions: How did Life come to be? If I stop to think about quarks and electrons merging to form neutrons and protons, which in turn join forces to create atoms and molecules, and maybe, just maybe, get ignited by electric charges or scorching temperatures, leading to the formation of biopolymers, cells, organs, and ultimately, the pinnacle of evolution—the human brain and consciousness itself—well, I can’t help but wonder: How did Life come to be? Get ready, dear reader, because today we’re diving headfirst into precisely that: quantum simulations of the primordial soup. Let’s go.

The Primordial Soup: Did Life Originate from Simple Molecules?
Diving into the origins of life is no small feat, my friend. It’s a grand pursuit that weaves together biology, chemistry, physics, astronomy, and geology. For the longest time, though, the quest for understanding life’s emergence and its potential existence beyond Earth belonged to the realms of religion and philosophy alone.
But then, in 1871, a spark of brilliance emerged from the depths of Charles Darwin‘s mind. In a letter to J. D. Hooker, he dared to propose a hypothesis, envisioning life blossoming in “a warm little pond, with all sorts of ammonia and phosphoric salts, lights, heat, electricity.” And just like that, the seed of the primordial soup concept was sown.
In the 1920s, Oparin and Haldane independently expanded upon this notion, crafting a comprehensive geochemical theory regarding the origins of life through the amalgamation of simple molecules like water, ammonia, and methane.
Fast forward about three decades, and Stanley Miller, under the supervision of Harold Urey at the University of Chicago, published a concise yet groundbreaking paper in the Science edition of May 15, 1953. Titled A Production of Amino Acids Under Possible Primitive Earth Conditions, Miller’s work revealed astounding results stemming from an electric discharge effect on a mixture of methane, ammonia, hydrogen, and, of course, water that recreated the primordial atmosphere as known at the time.
This is what’s commonly known as the prebiotic soup—pretty catchy name, right?
Miller used this sophisticated contraption with electrodes and other quasi-steam-punk gadgetry to make it happen. He sucked the air out of the system, added the gases, poured in some water, and let the electricity do its thing. After a week, he found this sticky goo at the bottom of the apparatus, and guess what?

The goo contained a significant amount of amino acids alongside hydroxy acids, short aliphatic acids, and urea. What made this even more astonishing was that the products weren’t a haphazard mixture of organic compounds; rather, a select few compounds emerged in surprisingly high concentrations. Even more awe-inspiring was the fact that these products held biochemical significance: they were amino acids, the very building blocks that form your proteins—the unsung heroes of the molecular world.
This feat of scientific ingenuity is now known as the Miller-Urey experiment—the primordial soup that breathed life into existence. With this groundbreaking discovery, a new era of origin-of-life investigations took flight, sparking our curiosity and propelling us further on this remarkable journey.
Now, if some of you, fellow adventurers, are itching to create amino acids from simple molecules, an educational version of this experiment can be carried out with some basic chemical equipment. But hey, hold your horses! Electric shocks, suffocating smells from ammonia, and dizziness-inducing fumes are no joke.
So, if you’re anything like me—a disaster waiting to happen in the lab—let’s opt for simulations instead. Way less chance of blowing something up or poisoning ourselves, right?
More on From Atoms To Words:
▸ From Earth to the Cosmos: How Hydrogen Bonds Shape Life
▸ Quantum Nanoreactor Simulations of The Early Universe
▸ Water’s Hydrogen Bonds: What Makes Them Vital for Life As We Know It?
Quantum nanoreactor simulations of the primordial soup
Some believe that the historical role of computational chemistry has been solely to describe chemical processes, rationalize mysterious phenomena, and settle experimental disputes. But let me tell you, my friend, things have taken a turn. We’ve witnessed some serious advancements in algorithms and computation in recent years, unleashing a whole new realm of capabilities. Now, simulations may be used not only to arbitrate different hypotheses, but also as a discovery tool to reveal unknown chemical mechanisms.
It’s game on, babe.
Right at the center of this momentous transition, we find the star of today’s story: the quantum nanoreactor. Oh yeah, this kickass method is grabbing all the spotlight in the community. We’ve already talked about it on From Atoms To Words when we delved into those mind-bending quantum simulations of the early universe. You remember that, right?
Let me refresh your memory. The quantum nanoreactor is this wicked technique that uses ab initio molecular dynamics to simulate the reactions of freely reacting molecules. You can set your molecular mixture and environmental conditions, such as temperature for example, and let the laws of quantum mechanics work their magic. Within this framework, you can then discover all sorts of reactions or stumble upon some never-before-seen reaction schemes.
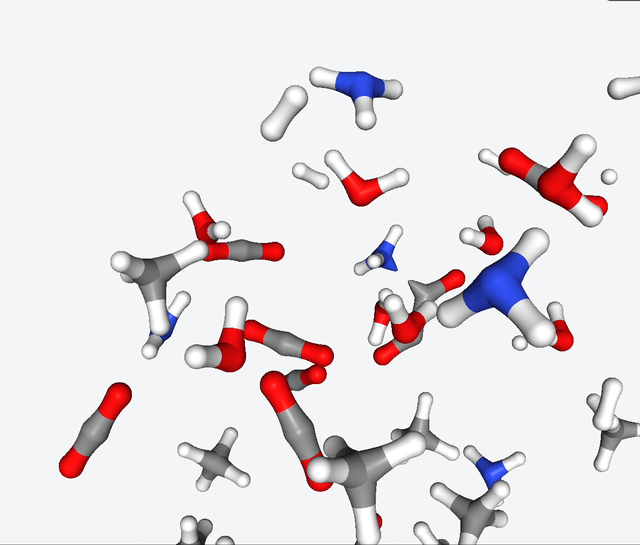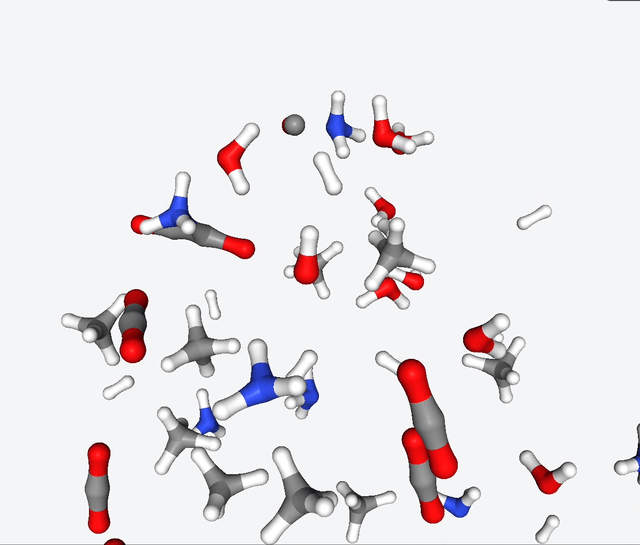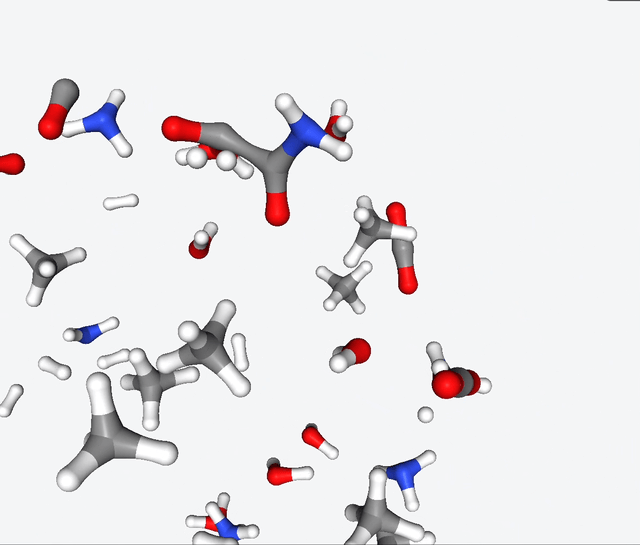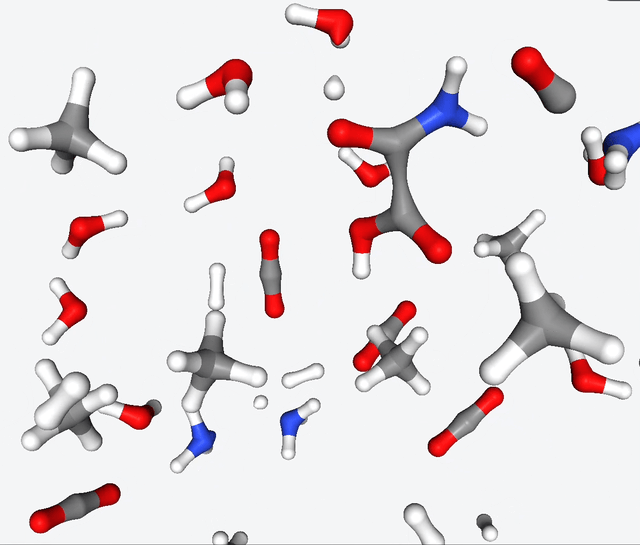
Now, Martinez & Co. had the brilliant idea of applying the quantum nanoreactor to the primordial soup. Yeah, you heard that right—they simulated the legendary Urey-Miller experiment. They replicated the conditions with hydrogen, ammonia, methane, water. They also added carbon monoxide to the mix, and ran several simulations with 50 to 100 reactants.
Hold on to your seat because the results are somewhere between pretty damn cool and totally mind-blowing.
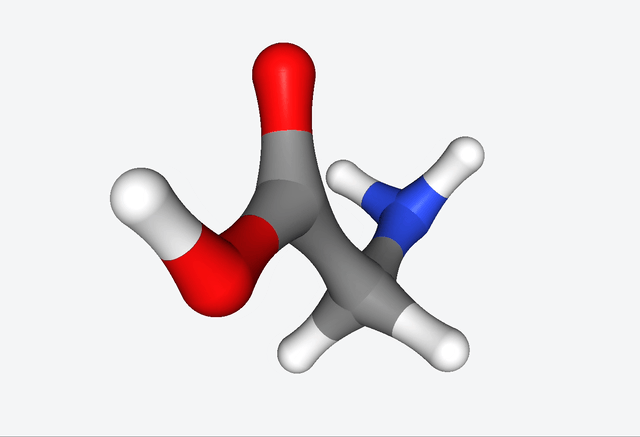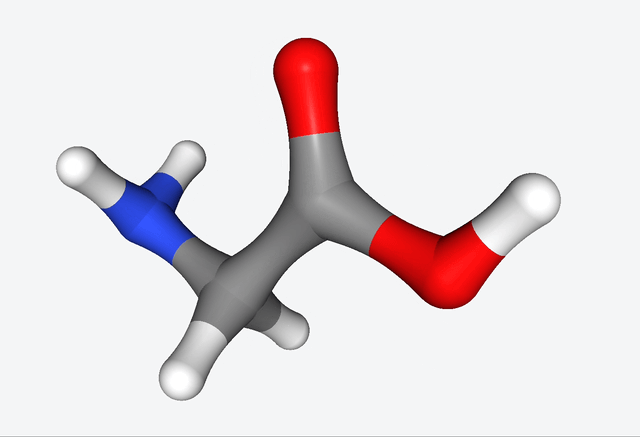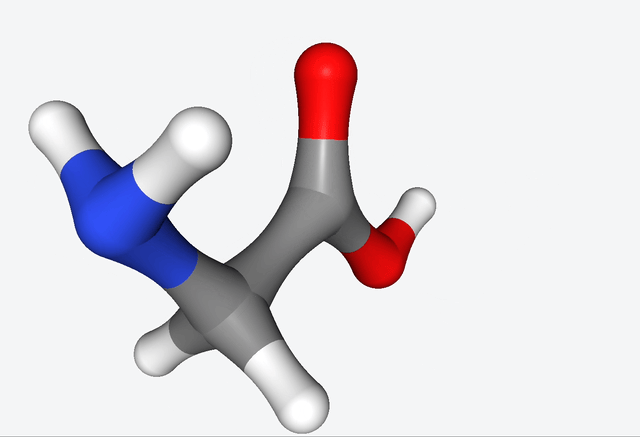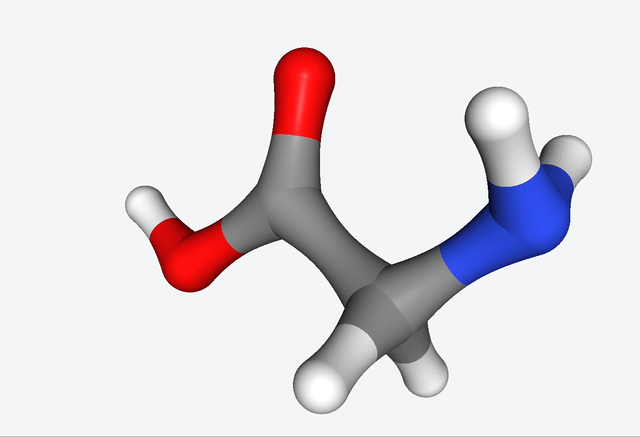
The simulations of the primordial soup produced a whole different bunch of molecules, like urea, ethylene glycol, and isocyanic acid, all of which have actually been found in meteorites that might’ve brought organic matter to Earth. But here’s the real kicker: Martinez & Co. also observed the formation of glycine, a natural amino acid, along with some funky non-natural variations like α-hydroxyglycine and α-aminoglycine, and a reduced form of alanine.
But wait, there’s more to this quantum nanoreactor simulation game. It’s not just about discovering new molecules. When you throw in some machine-learning routines, you can shed light on the most likely reaction pathways. In the case of the simulation of the primordial soup, for example, Martinez & Co. found over 700 connected reactions occurring during the Urey-Miller experiment.
To make sense of all this glorious chaos, they zoomed in on specific molecules in the reaction network and carefully analyzed the energy changes during their primordial reactions. And guess what? They stumbled upon several pathways leading to the formation of glycine. It’s like a chemical convergence, where all roads lead to Rome, with reaction barriers below 40 kcal/mol. This suggests that the reactions may be slow, but they have a good chance of occurring given enough time.

More on From Atoms To Words:
▸ The Evolution of Quantum Chemistry: From Pencil and Paper to Quantum Computing
▸ Quantum Chemistry of Molecule-Surface Adsorption: The 30-Year Struggle To Chemical Accuracy
▸ All-Atom Molecular Dynamics of SARS-CoV-2: The Computational Microscope’s View of 305 Million Atoms
Simulations of the Primordial Soup: Beyond the Miller-Urey Recipe
I know, I know. There are so many things to care about in this world. So, why are the quantum nanoreactor simulations of the primordial soup such a big deal that they deserve to be published in Nature Chemistry?
For starters, glycine is a superstar in the world of biomolecular building blocks. It’s the simplest amino acid and can be found in comets and meteorites. Studying how it’s formed in space, how it travels to us, and how it reacts to form biopolymers is a massive step towards understanding the origins of life itself.
But hold on, recent studies on prebiotic chemistry have thrown us a curveball. Turns out, the early Earth’s atmosphere might not have been as reducing as we thought, which means that the classic Miller-Urey mixture needs a little tweaking. This inspired a buffet of slightly different recipes for the primordial soup, each with its own unique flavor. Maybe we need to throw in some nitrogen, carbon monoxide, and heck, even a dash of oxygen. And here’s where the quantum nanoreactor, and atomistic simulations more generally, may come in handy: to reestablish the historical role of computational chemistry in rationalizing experimental results and predicting previously unforeseen reactivities.
But let me tell you, my friend, it’s not only about these wild primordial soups. The quantum nanoreactor is a game-changer for all kinds of chemical applications. We’re talking about predicting reactivity, untangling those tricky degradation paths, and unveiling the intricate webs of reaction networks that matter in the world of industry and academia.
It’s like having the ultimate chemistry toy in our hands, and let me assure you, my colleagues at Quantistry and I are having an absolute blast with it, applying it to a variety of molecular systems.
More on From Atoms To Words:
▸ ReaxFF Molecular Dynamics: Simulating Complexity Beyond Quantum Chemistry
▸ Digital Alchemy: Computers in Chemistry and the Future of Scientific Discovery
▸ Multiscale Simulations of DNA: From Quantum Effects To Mesoscopic Processes
A Final Personal Touch
This primordial soup story drives me absolutely nuts, man. I mean, seriously, if you take a moment to really wrap your head around it, that whole experiment is mind-blowing.
Picture this: Miller throws a few molecules together, zaps them with electricity, and bam! After just a few days of running the experiment, he ends up with this gooey concoction loaded with amino acids. It blew everyone’s freakin’ mind. People at the time believed that if they had let the experiment go, some living creatures would start crawling out of that lab!
But you know what life does? It sends you lemons, man. And guess what? No creatures crawled out of any lab. Total bummer, right?
The thing is, modern cells have been through billions of years of evolution and some scientists argue that we need to fully understand life as it exists right now before we can even think about figuring out how it all started. But here’s another way to look at it: maybe it’s by trying to understand the transition from non-life to life that we’ll actually get a grip on the most fundamental properties of life itself. Maybe that’s where the real juicy secrets are hidden.
So tell me, my dear reader, what’s your take on it? Do you think we’ll ever witness some weird creatures crawling out of a primordial soup experiment?
Well, one thing is for sure: it’s gonna take some real quantum computing power to simulate that. But hey, who knows? The universe has a sneaky habit of always finding ways to surprise us.
If you enjoyed this dive into the simulations of the primordial soup, I’d love to hear your thoughts. Agree, disagree, or have a totally wild theory of your own? Let’s connect! Subscribe to my LinkedIn newsletter and let’s keep the conversation rolling.
Scientific Reading List:
▸ The 1953 Stanley L. Miller Experiment: Fifty Years of Prebiotic Organic Chemistry 2003
▸ Discovering chemistry with an ab initio nanoreactor 2014
▸ Re-conceptualizing the origins of life 2017
▸ The Prebiotic Kitchen: A Guide to Composing Prebiotic Soup Recipes 2021
▸ Prebiotic chemistry and origins of life research with atomistic computer simulations 2020