As a student, I used to turn my nose up at the quantum chemistry of materials, polymers, or molecule-surface adsorption. My interest lay solely in the chemical processes of biological systems. I mean, there is something undeniably alluring about the mysteries of DNA. Since I joined the team at Quantistry, my perspective has shifted. I’ve been exposed to a whole new world of material science, and, well, it’s utterly captivating. So, today, I want to take you on a journey through the scientific history of how we have reached chemical accuracy in the prediction of the adsorption energy of carbon monoxide on magnesium oxides, or as the scientists like to call it: CO/MgO. Just like the battle of theory vs. experiment to estimate the hydrogen dissociation energy, the long and controversial saga of attempts to predict the CO adsorption energy on MgO surfaces is an epic tale of quantum chemistry, molecule-surface interactions, and the unyielding pursuit of scientific understanding. There is a story hidden here. Let’s uncover it together.
Quantum Chemistry of Molecule-Surface Adsorption: The Hard Case of CO/MgO
Let’s give a shout-out to the stars of surface science: oxides. These surfaces are the result of metal cations and oxide anions creating some essentially cool chemistry. We’re talking about thermal coating, corrosion protection, and microelectronics. Thin films of oxides are crucial in everyday things like computer chips and other devices. The ultimate holy grail of surface chemistry is to harvest the sun’s energy via photocatalysis, photoelectrocatalysis, and dye-sensitized solar cells.
Now, let’s get to the nitty-gritty: molecule-surface adsorption on oxide surfaces — a process at the heart of the most important applications of oxides. Heads up: Adsorption is a real beast to tackle.
Enthalpy, entropy, and a whole host of complex reaction networks and potential surface sites, no wonder that Wolfgang Pauli — yes, the Pauli exclusion principle dude — once said that surfaces were “invented by the devil.”
But fear not, because our beloved quantum chemistry has some serious tools up its sleeve. This time, thanks to none other than Prof. Sauer from the Humboldt University of Berlin. Now, Prof. Sauer is not your run-of-the-mill computational chemist. He’s a bit of a celebrity in Germany. Not only is he married to Angela Merkel (who, by the way, earned a PhD in quantum chemistry — perhaps that’s why she has been such a successful leader), but Prof. Sauer is also the recipient of the Schrödinger Medal for “his outstanding contributions to the quantum chemistry of solid materials.”
We will go much more into the details of it, but — spoiler alert! — his recent theoretical approach has enabled a serious advancement in the prediction of molecule−surface adsorptions, reproducing available experimental data within chemical accuracy (<1 kcal/mol) — the gold standard of quantum chemistry.
Such a major milestone didn’t happen overnight. It’s the result of decades of painful experimental work and careful theoretical development, conflicts, and disagreements. And it all started in the late 60s with the adsorption of carbon monoxide on magnesium oxide surfaces, the focus of our story today: CO/MgO.

More on From Atoms To Words:
▸ Predicting The Hydrogen Dissociation Energy: The 100-Year Battle of Quantum Chemistry vs. Experiment
▸ The Evolution of Quantum Chemistry: From Pencil and Paper to Quantum Computing
▸ Water’s Hydrogen Bonds: What Makes Them Vital for Life As We Know It?
Quantum Chemistry of Molecule-Surface Adsorption: The early studies
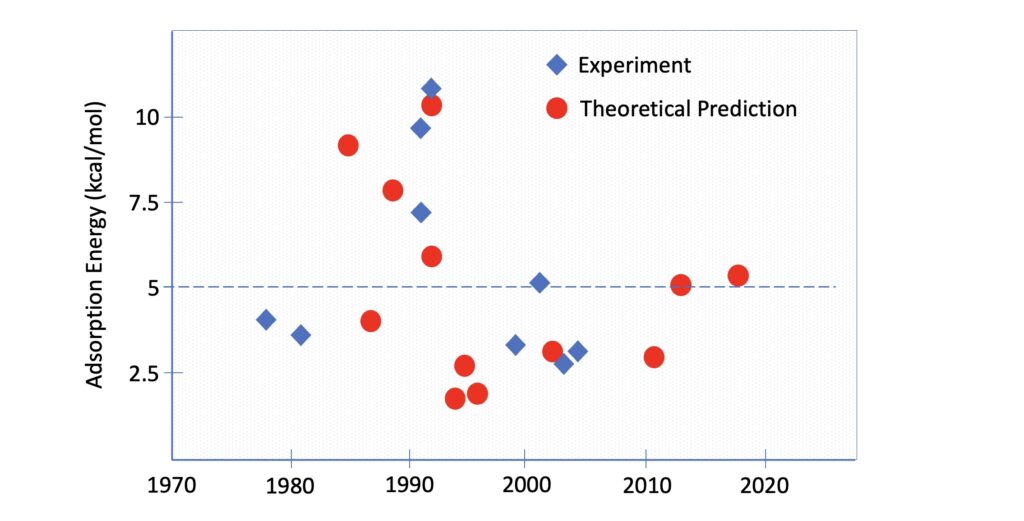
Let’s take a trip down memory lane to the groovy year of 1966, when The Beatles were embarking on their third and final tour of North America, while Lunsford and Jayne were breaking ground in the field of surface science. These pioneers discovered that the adsorption of CO on MgO created a paramagnetic species and a unique signal in ESR experiments, paving the way for future research on the topic. Move ahead to the late 70s and early 80s, when Furuyama and Pauskshtis carried out more experimental work and quantified the adsorption energy of CO on MgO powders to be in the range of 3.5-4.0 kcal/mol. A relatively poor binding, comparable to weak hydrogen bonds.
Pretty hectic experimental activity, but what about quantum chemists? Well, our quantum friends didn’t have time for mindlessly watching their favorite TV shows either.
In the early 80s, while Manin and Feynman were deep in thought, contemplating the use of quantum computing to describe the quantum mechanical nature of physical and chemical systems, Colbourn and Mackrodt proposed the first ab initio calculations of CO adsorption on MgO. Armed with their cutting-edge all-electron Hartree-Fock calculations (which, let’s be honest, were rather raw compared to today’s computational standards), they predicted that CO likes to bind at the Mg2+ sites with the carbon end down, with an adsorption energy of 9 kcal/mol.
Dovesi & Co. confirmed a slight preference for the carbon end down arrangement, but estimated an adsorption energy of only 4 kcal/mol, consistent with the experimental findings. A couple years later, the same computational approach yielded an even lower adsorption energy of 2 kcal/mol for a full monolayer coverage (one CO molecule on each Mg2+ ion).
So, the theoreticians took a crack at it and good news: they were in the same ballpark as the experimental results.

The dark 90s: does quantum chemistry work at all for molecule-surface adsorption?
Let’s dive into the 90s, when researchers made some serious strides in understanding the behavior of carbon monoxide on magnesium oxide surfaces. Two experimental studies, one by Henry and the other by He, estimated the adsorption energy to be 9 kcal/mol, in agreement with the early calculations by Colbourn and Mackrodt.
But hold on, what about Dovesi & Co. and the earlier experiments? Which one of the two sets of values is correct, 4 or 9 kcal/mol?
Some hope to solve the matter came from the theoretical front; while up to now all calculations relied on wavefunction methods, in 1992, the first theoretical studies using Density Functional Theory (DFT) emerged. This more efficient quantum mechanical method, based on electron density rather than wavefunctions, raised expectations and created quite a buzz. But, as is often the case with new and exciting things, the enthusiasm didn’t last long. Neyman and Rösch burst onto the scene with an eye-popping estimate of 22 kcal/mol for the CO/MgO interaction — way too high compared to both experiments. It was a major letdown for DFT, but fortunately, the theoretical chemists persisted.
Nygren & Co. took a more accurate approach, which included a refined description of the electrostatic potential of the surface, correlation effects, and BSSE corrected energy. Despite all these improvements, their final estimation was way too low, in the range of 2 kcal/mol. A year later, Mejias adopted a similar computational scheme, but the highest level of computation so far predicted an even lower value, only 0.2 kcal/mol. Yup, that’s barely an interaction at all.
It was a classic case of one step forward, two steps back. Theoretical chemists just couldn’t seem to catch a break. But for a brief moment, it seemed that a break was going to be caught.
As the 90s came to a close, all hope seemed lost in understanding the behavior of carbon monoxide on magnesium oxide surfaces. But then, like a phoenix rising from the ashes, a new theoretical study by Chen & Co. reignited the flame of possibility. Using a band structure periodic model and plane waves, they meticulously computed the adsorption energy to be 6.5 kcal/mol. It wasn’t perfect, but it was a step in the right direction towards the latest measurements of 9 kcal/mol.
But, theoreticians, man, are a weird bunch. Always chasing, never happy.
Just when it seemed like they were making some progress, the community of quantum chemists came in hot, raining down objections. Several authors published a comment, and then Chen had to reply. Drama! The commenters reminded everyone that all previous theoretical studies had painted a completely different picture of the interaction process, with much lower estimates of the adsorption energy, no matter what method was used.
This was no petty disagreement — Quantum chemists were tearing their hair out over this one.
More on From Atoms To Words:
▸ Computational Chemistry 2043: A Quantum Peep into the Future
▸ Digital Alchemy: Computers in Chemistry and the Future of Scientific Discovery
▸ Can Quantum Chemistry Simulations Help Trace the Origin of Life?
The New century: experiments and calculations converge
The theoretical results just weren’t lining up. It seemed that the more quantum chemists refined their calculations, the smaller the energy became. So, naturally, CO adsorption became the topic of the day. Even non-specialists were getting in on the action and theoretical studies of molecule-surface adsorption were scrutinized like never before.
The question on everyone’s minds was a doozy: does quantum chemistry work at all for molecule-surface adsorption?
To put the matter to rest, Freund & Co. set up a meticulous measurement, using thermal desorption spectra. They estimated the adsorption energy to be 3.2 kcal/mol. This was close to the early experiments, but provided a very different picture from the most recent ones.
But even with this breakthrough, the debate raged on. An entire community in the dark! The experimental and theoretical techniques, although improving over the years, seemed to diverge on the estimation of the adsorption energy of such a “simple” system.
Did experiment and theory yield the same physical quantity? Did experiment and theory look at the same surfaces?
As we headed into the new millennium these questions were forgotten as the debate over the adsorption energy of carbon monoxide on magnesium oxide surfaces seemed to come to a close. More and more refined experimental and theoretical studies continued to emerge left and right, eventually converging on an estimated adsorption energy of 2-3 kcal/mol.
Quantum Chemistry reaches Chemical accuracy for Molecule-Surface Adsorption
Just when everyone thought the matter was settled, someone came along and shook things up. That someone was Prof. Sauer. Remember? The hero of our story. In 2013, Prof. Sauer published the first of a series of articles that would turn the world of adsorption energy on its head.
Using a robust hybrid quantum chemical method (conceptually similar to a previous study), Prof. Sauer’s approach blended the best of two worlds: a high-level wave-function for the “reactive center,” and a more efficient DFT approach to model the rest of the oxide. These two layers were then combined via a subtractive scheme, in an approach similar to Morokuma’s well known ONIOM.
So, here’s the deal: Prof. Sauer did some heavy-duty number crunching and came up with an adsorption energy of 5 kcal/mol. Sounds pretty good, right? The thing is, it wasn’t quite up to snuff with the generally accepted value of 2-3 kcal/mol.
But here’s where the story gets interesting.
Instead of throwing in the towel, Prof. Sauer went ahead and submitted his findings anyway. And thank goodness he did because during the submission process an eagle-eyed reviewer pointed out an overlooked experimental study by Dohnálek & Co, who had reported an adsorption energy of 4.4 kcal/mol, which, considering the experimental error, aligned very well with Prof. Sauer’s calculations.
The real kicker was that Dohnálek & Co revisited the data analysis from previous measurements and demonstrated that the pre-exponential in the Redhead equation, typically used to derive the experimental energies, was off. They adjusted the pre-exponential factor to 1015±2 (instead of the usual 1013), and voila — the previous measurements suddenly made sense, and everything converged around an adsorption energy of 5 kcal/mol.
Encouraged by this, Prof. Sauer went back to the drawing board, fine-tuned his calculations, and obtained a final theoretical estimation for the CO/MgO adsorption energy within 0.5 kcal/mol from the Dohnálek’s experiment.
And that, my friends, is how you solve a 30-year-old puzzle! It took years of hard work and persistence. Prof. Sauer believed in his approach and dared to dare. It paid off in spades.
Chemical accuracy in quantum chemistry: From Metal Organic Frameworks to CO2 reduction
Chemical accuracy has been achieved for real and significant catalytic processes. We now have procedures that will enable the calculation of reaction rates for heterogeneously catalyzed reactions within experimental error.
Richard Catlow
You might think that Prof. Sauer would rest on his laurels after achieving such a mind-blowing feat, but nope — the man continued to rock. With his team, he decided to take their successful approach and apply it to other molecule-surface systems, from MgO surfaces to metal-organic frameworks (the famous MOFs) and zeolites interacting with H2, N2, CO2, CH4, and C2H6. And you know what? They absolutely nailed it, reaching once again the quantum chemistry gold standard of chemical accuracy (<1 kcal/mol).
The cool thing is, others took notice and employed Prof. Sauer’s approach for other molecule-surface interactions. One such example is the 2023 study by Chen & Co. published in Nature. They achieved near chemical accuracy in describing copper-based heterogeneous catalysis, a key process that holds a great promise for the reduction of CO2 into valuable chemicals and fuels.
Paraphrasing the words of Richard Catlow: Hot damn, we’ve done it! We’ve cracked the code of computational modeling and reached the point of chemical accuracy for catalytic processes that actually matter.
What an exciting time to be a quantum chemist.
More on From Atoms To Words:
▸ Quantum Biology: The Fuzzy Connection Between Quantum Mechanics and Living Things
▸ Let’s Fight Climate Change With The Computational Design of Metal-Organic Frameworks (MOF’s)
▸ Chemical Space to Material Discovery: Simulations and Machine Learning Leading the Way
A Final Personal Touch
Just like the saga of the hydrogen dissociation energy, our story today about the quantum chemistry of molecule-surface adsorption is a perfect example of how scientific insight grows in our modern era — with lots of twists, turns, and controversy along the way.
Through all of these challenges, we’ve learned that the pursuit of knowledge is never a straight path, but it rather emerges from the confrontation of theory and experiment, from different ideas and points of view clashing, merging, converging.
So, let’s embrace the battle of theory vs. experiment, for it’s a never-ending journey that ultimately leads to a better understanding of our world.
If you enjoyed this dive into the quantum chemistry of molecule-surface adsorption, I’d love to hear your thoughts. Agree, disagree, or have a totally wild theory of your own? Let’s connect! Subscribe to my LinkedIn newsletter and let’s keep the conversation rolling.
Disclaimer: I have to admit that this story was hard to write. From the early studies up to the most recent works of Sauer and Chen, this stuff is quite technical. I did my best to report it as accurately as possible and to make it a fun read. For a rigorous review on the topic, I encourage you to delve into Sauer et al.’s original paper.
Scientific Reading List:
▸ Ab Initio Calculations for Molecule−Surface Interactions with Chemical Accuracy – Sauer 2019
▸ Prediction of Rate Constants for Catalytic Reactions with Chemical Accuracy – Catlow 2016
▸ Accurate adsorption energies of small molecules on oxide surfaces: CO–MgO(001) – Boese and Sauer 2013
▸ Quantum Chemistry of Oxide Surfaces: Pacchioni 2000