It all began with a project on cisplatin at the University of Leiden in the Netherlands, under the expert guidance of Prof. Reedijk. Now, Prof. Reedijk’s team was also at the forefront of pioneering experimental research in anion-pi interactions, and that’s the beauty of scientific collaborations—they lead you down unexpected paths. Our collaboration was no exception. It soon blossomed into a study I hold in high regard, one of the earliest predictions of the role of the anion-pi interactions in proteins. Since those early days, I’ve watched this field grow to influence diverse areas of chemistry, including organic electronics, supramolecular assemblies, and catalysis. Today, I’m excited to take you on a deep dive into anion-pi interactions, exploring their nature, their functioning, and the broad impact they are having across chemistry. Ready? Let’s go.

ANION-PI INTERACTIONS: THEORETICALLY PREDICTED, EXPERIMENTALLY CONFIRMED
Rewind to 2004, a landmark year in supramolecular chemistry. It was when the first experimental evidence of anion-pi interactions made its debut, revealing the attraction between anions and electron-poor aromatic rings. This was unexpected, as anions are negative and aromatic rings are loaded with pi electrons.
Quite counterintuitive, right?
This breakthrough, led by my later collaborator Gamez within Prof. Reedijk’s team, didn’t just come out of the blue. The theoretical groundwork was laid back in the late 90s. Quantum chemists used methods such as Hartree-Fock, MP2, and DFT calculations to predict the binding between electron-rich groups and aromatic rings like hexafluorobenzene or triazine.

What sets these interactions apart is their distinctive nature, primarily driven by electrostatics. Despite being noncovalent, they carry a noteworthy energetic impact, typically ranging between 2 and 16 kcal/mol. They may not be the strongest players, but they have a knack for subtly influencing chemical processes.
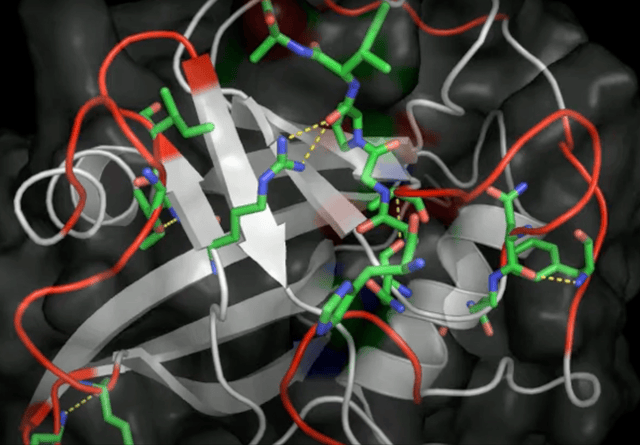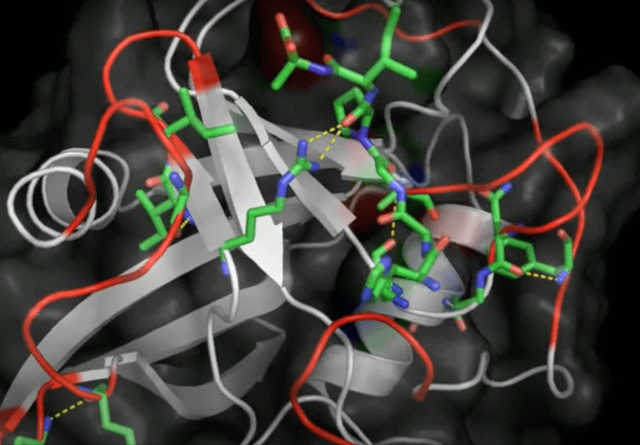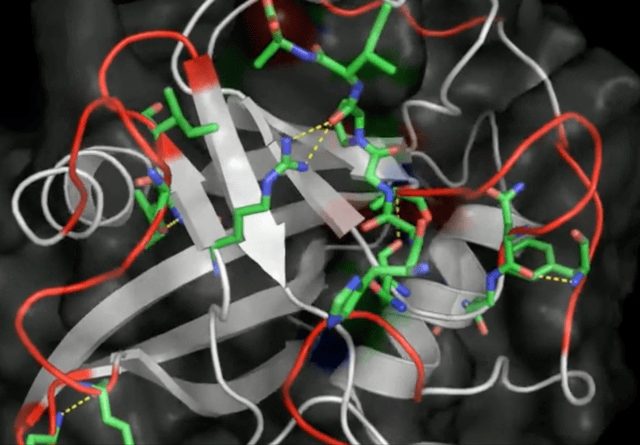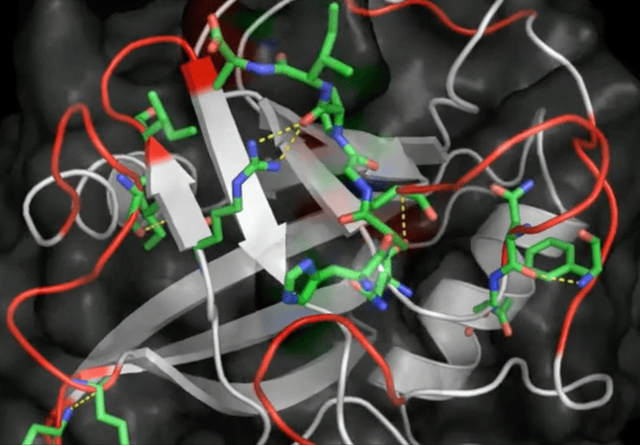
Initially seen as a novelty, this type of noncovalent bonding caught wider attention, especially in biochemistry. It was the year 2011 and I was right there in the thick of it, diving into the Protein Data Bank and publishing the first evidence of anion-pi interactions in protein structures. What did I find? Aromatic amino acids—tryptophan, phenylalanine, tyrosine, and histidine—in close contact with anions, such as chloride and phosphate groups.
As of today, anion-pi interactions are gaining fame, with research expanding our understanding across various domains—from supramolecular chemistry to anion channels, proteins, and DNA/RNA structures.
But why should we care about these interactions?
Well, because anions are pretty much everywhere in nature. Almost every conceivable biochemical activity involves the recognition and transport of anions at some level. Not to mention numerous chemical processes.
So, are you curious about which corners of chemistry are really feeling the impact of anion-pi interactions?
Further reading: Recent advances in anion-pi interactions | Robertazzi, 2011
More on From Atoms To Words:
▸ 7 Noncovalent Interactions in Proteins: The Hidden Architects of Structures and Functions
▸ The Lifesaving Hunch: How Rosenberg’s Unexpected Discovery of Cisplatin Changed Medicine
▸ The Evolution of Quantum Chemistry: From Pencil and Paper to Quantum Computing
ANION-PI INTERACTIONS: FROM METAL-ORGANIC FRAMEWORKS TO ION TRANSPORT PROCESSES
What started off as just a hunch in theory, later confirmed by some nifty experiments, anion-pi interactions are now a big deal in the chemistry world, from the inner workings of metal-organic frameworks to shedding new light on organic electronics, catalysis, and ion transport processes.
So, let’s dive deeper and explore what’s driving the excitement about these interactions in areas such as:
▸ METAL-ORGANIC FRAMEWORKS (MOFs)
You might recall our conversation on MOFs from earlier posts on From Atoms To Words. These beautiful megastructures, where metal ions are linked with organic molecules, boast exceptional porosity and versatility. Their unique design allows MOFs to excel in a variety of areas, including carbon capture and conversion. A key to their success lies in anion-pi interactions, as shown by Domasevitch’s team. They found, for example, that oxygen atoms in perchlorate and pyridazine rings within certain MOF structures do engage in anion-pi interactions. Shifting focus to responsive materials, consider MOFs that exhibit photochromic behavior. These materials have the ability to change color upon light exposure. Here, anion-pi interactions are pivotal, enabling quick and reversible color transitions. This feature is highly valuable in the development of advanced photoswitches and sensors. But their influence in MOFs doesn’t end there. Saha’s team’s findings revealed another intriguing aspect: these interactions can significantly boost lithium-ion conductivity. This underscores their diverse role, not just as a structural element but also as a functional enhancer, expanding the capabilities of MOFs in a variety of applications.
▸ ORGANIC ELECTRONICS
Organic electronics, a branch of materials science focused on organic molecules with impressive electronic properties like conductivity, is witnessing a game-changing influence. And the spotlight-stealer? Anion-pi interactions. Their noncovalent nature is essential for boosting complex electronic communication and keeping molecular structures stable. Take Matile and team’s work, for example. They made a significant leap by increasing pi-acidity in naphthalenediimides, leading to innovative chiral redox switches. This finding is a prime example of the power of anion-pi interactions in constructing sophisticated and efficient electronic devices. Another cool case is light-induced molecular switches and organic solar cells. Here, this kind of interactions are crucial for achieving high conductivity and enhancing conversion efficiency in materials like fullerene ammonium iodide. In energy storage systems, these interactions are integral to the improved efficiency of lithium organic redox flow batteries. The big picture? The study of these interactions within organic electronics is setting a new course for the industry, ranging from renewable energy solutions to sensing technologies.
These results show for the first time that anion-pi interactions may play also a role in proteins, opening the way to further experimental and theoretical studies, which may shed light on those biological processes in which anions are involved and for which anion-pi interactions may be relevant.
Robertazzi 2011
▸ CATALYSIS
Catalysis, the process where a substance known as a catalyst speeds up a chemical reaction without being consumed, is a cornerstone of modern industry. It’s everywhere, from water treatment to fuel cells and green synthesis of fine chemicals. Remember Matile and team’s work on organic electronics? They’ve been exploring anion-pi interactions in transmembrane anion transport too, bringing a fresh perspective to catalysis. Their approach? Focusing on stabilizing anionic transition states on pi-acidic aromatic surfaces, which effectively lowers the activation barriers of reactions. But it’s not just about speeding up reactions. Anion-pi catalysis is about precision and efficiency. It opens up new possibilities for more selective, faster, and potentially more environmentally friendly catalytic processes. And that’s not all—it’s paving the way for innovations across a myriad of industries, from pharmaceuticals to renewable energy.
▸ Ion Transport Processes
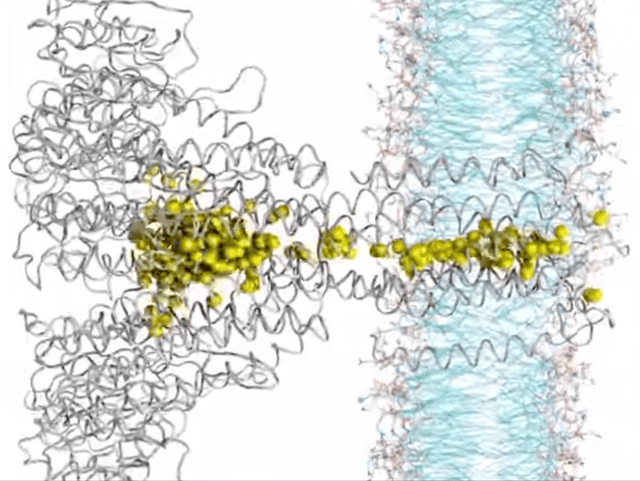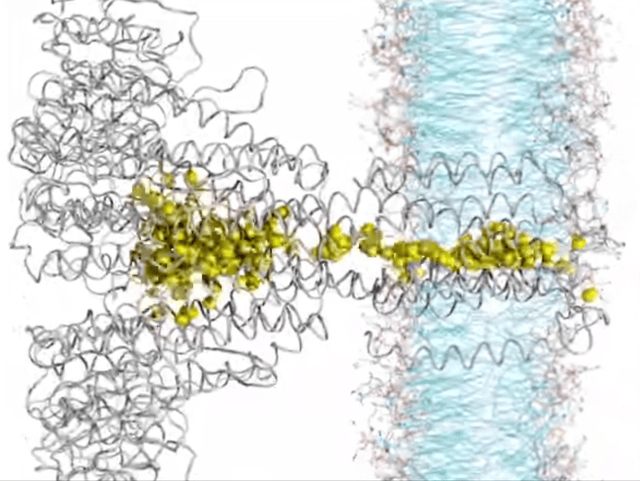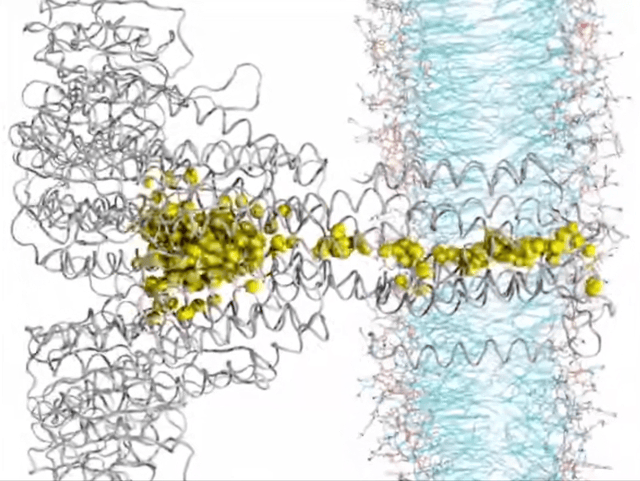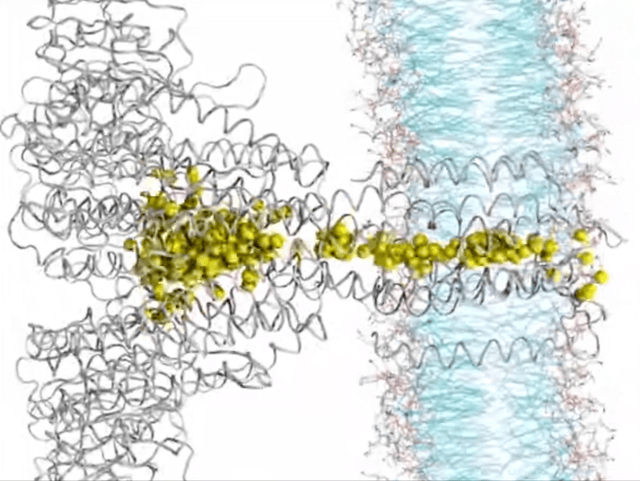
Here’s where things get really interesting. Ion transport, in a nutshell, is all about how ions hustle across membranes, playing a crucial role in the nitty-gritty of cell life. Think nerve signals zipping, muscles contracting, or keeping our body’s electrolytes in check. Traditionally, this selective ion transport in biological systems has been driven by noncovalent interactions like hydrogen bonding, dipole interactions, or ion pairing. But now there’s a new player in town—yes, I am talking about anion-pi interactions. These interactions are reshaping our understanding of how ions make their way through biological membrane channels. It turns out that receptors with milder anion-pi interactions are outshining those with the stronger halogen or hydrogen bonds, showing higher ion transport activity. This growing research area is highlighting the unique role of anion-pi interactions in boosting both the efficiency and specificity of ion transport. Focusing on anion-pi interactions is a move that promises a lot, pointing us toward a better understanding of biological ion transport.
▸ Systems of Biological Relevance
The field where anion-pi interactions really shine is in life’s molecular mechanisms. For example, Frontera’s computational body of work showed that these interactions are key to how enzymes like urate oxidase function, highlighting their vital role in biological regulation. The pharmaceutical world is also taking notice, especially in the development of antituberculosis drugs. Researchers have spotted these interactions in the way specific inhibitors bind to enzymes, opening doors to new therapeutic approaches. One particularly thrilling aspect? The role of anion-pi interactions in the voltage sensing of certain motor proteins, like prestin. This insight is a game-changer for bioelectronics, showing how these interactions can lead to electromechanical conversions.
Further reading: Emergence of anion-pi interactions: The land of opportunity in supramolecular chemistry and beyond | Ali, 2020
More on From Atoms To Words:
▸ Serendipity in Science: What’s its Fate in the Age of AI?
▸ Machine Learning in Materials Science: A Second Computational Revolution?
▸ Computational Chemistry 2043: A Quantum Peep into the Future
A final personal touch
Wow, can you believe it’s been almost twenty years since I first dipped my toes into the world of anion-pi interactions? Research that I started in Trieste at the International School for Advanced Studies (SISSA), continued in Cagliari, and finalized in Berlin—Ah, time flies when you’re enjoying the mysteries of chemistry!
Back then, these interactions were like uncharted territory, but now, they’re key players in a whole range of fields, from ion transport to the nifty gadgets in organic electronics, not to mention their role in catalysis and biological processes.
But let’s be real here. Sure, there’s a ton of theoretical and computational work out there, but when it comes to hands-on, experimental studies, we’re playing catch-up. To date, experimental reports of anion-pi interactions, both in solid-state and solution-phase, remain limited.
The silver lining? As Rashid Ali points out in his recent review article, anion-pi interactions are a land of opportunity, ripe for further exploration.
So, here’s a shout-out to all you curious minds and fellow researchers—there’s a treasure trove of discoveries waiting. Let’s roll up our sleeves and dive in, shall we?
If you enjoyed this dive into the role of anion-pi interactions in chemistry, I’d love to hear your thoughts. Agree, disagree, or have a totally wild theory of your own? Let’s connect! Subscribe to my LinkedIn newsletter and let’s keep the conversation rolling.