Back in my freshman year of university, sitting in a chilly chemistry lecture hall, I remember our professor passionately describing the extraordinary properties of water. There it was, a seemingly mundane substance, filling our glasses, covering vast expanses of the ocean, and falling as rain from the skies. At that time, I recall coming across a story where Enrico Fermi supposedly claimed that the peculiar behavior of water – decreasing its density when it freezes – was the ultimate proof of God’s existence. This unique trait of water, allowing life to thrive in the oceans, thus enabling life on Earth, seemed almost too perfect, too intentional. That Fermi quote? Well, it doesn’t exist. And yet, it’s true that the remarkable, sometimes even anomalous, properties of water are intricately linked to life as we know it. The source of this magic? It all boils down to water’s hydrogen bonds and their specific strength. So, how does this microscopic detail make such a colossal impact? Keep reading, ’cause that’s the heart of today’s story. So, grab your favorite drink (water, perhaps?) and let’s go.
The Essence of Water: More Than Just H2O
Have you ever stopped to think about the water you drink, the rain that falls, or the ice that chills your Aperol Spritz? It’s not just H2O.
There’s a cosmic quest unraveling far beyond our blue skies, with scientists discovering an abundance of water in our Solar System that dwarfs Earth’s supply. We’re talking about a whopping 32.5 to 65 billion cubic kilometers of the stuff. That’s right, up to 50 times the water twirling around in our oceans, rivers, and teacups.
But what’s the deal with all that water out there? It throws open the doors to the possibility that life might be sprinkling its magic in territories far beyond our own. Ice and vapor are pretty common across the universe. Yet, the star performer, liquid water, well, that’s a bit more elusive.
And that brings me to today’s big question: why is water so central to life?
Water serves as a medium for the transformation of highly complex organic molecules that form the basis for life processes. The reason for many of liquid water’s special properties originates from the water molecules consisting of dipoles that associate through intermolecular hydrogen bonds in its condensed phase.
F. H. Frimmel | 2007
Water molecules might seem like the understudies in the grand play of the universe, but they’re actually headlining the show. Water molecules pull off stunts that leave other liquids green with envy.
If you think about it, water is kind of a rebel in the chemistry world. It doesn’t follow the crowd. If water acted like other liquids, blanketing 71% of our planet, we might not have the life we’re familiar with today.
So, let’s bring the spotlight back home for a second.
Water is the only inorganic compound that naturally exists in three phases – solid, liquid, and gas – at the temperatures normally found on Earth. Water is often called the universal solvent. It’s the medium where the complex interactions of biomolecules weave the story of life’s processes.
And the key player in this ensemble? The hydrogen bond.
Further reading: The Physicochemical Properties of Water and their Relevance for Life, 2007
For Life’s Sake: The anomalous properties of water
On From Atoms To Words, we’ve already explored the power of hydrogen bonds in the works of catalytic and genetic molecular machines, our proteins and DNAs, but before we go deeper into the chemistry of it, let’s first dive into some of the unique properties of water that make it essential for life:
- Wide Liquid Range Temperature: Water remains liquid from 0°C to 100°C at sea level, offering a hospitable range for life far wider than that of other solvents like methane or ammonia.
- High Surface Tension: The strong attraction among water molecules at the surface results in a high surface tension, a vital trait for many life forms and plant survival mechanisms.
- Anomalous Density: Unlike most substances, water’s density decreases when it freezes, causing ice to float and providing an insulating layer that protects underwater life in harsh climates.
- Robust Hydrogen Bonds: Oxygen’s strong electronegativity in water molecules leads to robust hydrogen bonding, enabling a broader temperature range for liquid water, crucial for life’s diversity.
You know, these unique qualities of water aren’t just random quirks; they’re crucial to the whole story of life.
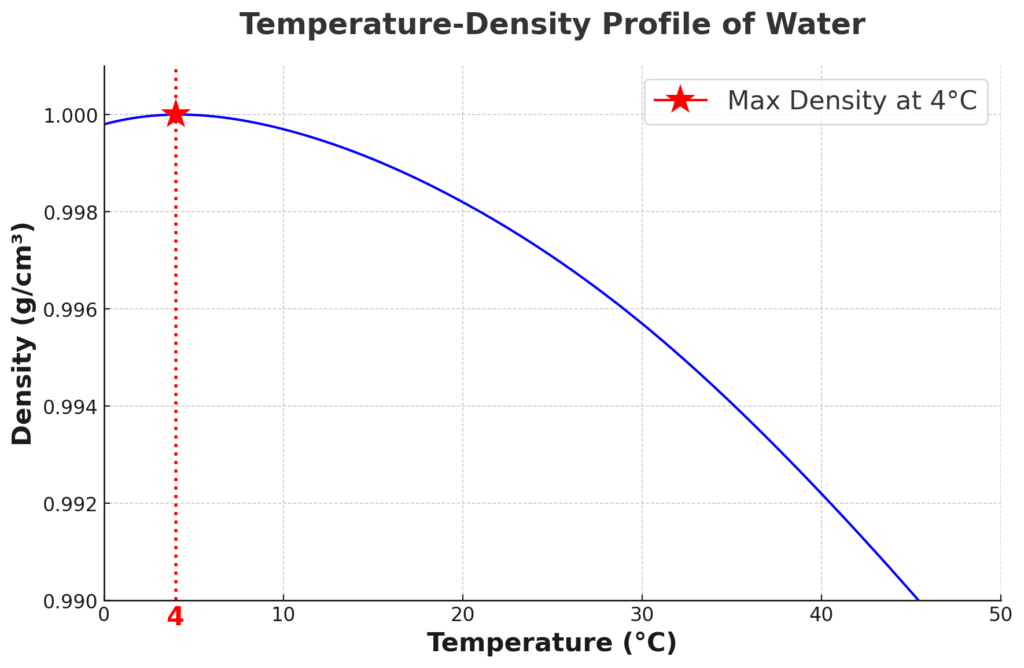
Let’s mull over this: water exhibits its highest density at 4°C. That means that water does something pretty unusual at this temperature – it expands, whether cooling down or heating up. This anomalous behavior, exclusive to water, has a bunch of ripple effects in nature. With water most dense at 4°C and ice less dense, lakes and rivers freeze top-down, protecting underwater life and circulating nutrients below.
Imagine if ice did the exact opposite and sank. We’d have a whole different scenario in winters, right? Instead of forming a frosty blanket on top, ice would plunge to the bottom, turning lakes and rivers into frozen graves for all the living things down there.
And let’s not forget about water’s broad temperature range in its liquid state. This range is like a big, welcoming embrace for life, stretching from the hot, humid tropics to the freezing poles. It’s water’s ability to store and move heat around that shapes our planet’s climates, influencing how and where different species have evolved over time. And on the molecular level, water’s a superstar solvent, especially for polar and ionic compounds, thanks to its polarity, high dielectric constant, and small size.
Water’s also a big deal when it comes to hydrating biomolecules such as proteins and nucleic acids, and thus determining how they function and form.
From the tiniest atomistic level to the vast size of the oceans, water is pretty much the lifeblood of our planet. It’s so woven into the essence of life that without it, the story of life here, there and maybe everywhere would be left untold.
So yeah, water is a big deal. And who’s the secret player behind all these amazing feats? That’s right, the hydrogen bond.
Further reading: Water’s Hydrogen Bond Strength, 2007

More on From Atoms To Words:
▸ 7 Noncovalent Interactions in Proteins: The Hidden Architects of Structures and Functions
▸ Modeling the Origins of Life: Quantum Simulations of the Primordial Soup
▸ Quantum Biology: The Fuzzy Connection Between Quantum Mechanics and Living Things
Water’s Hydrogen Bonds: Tailor-Made For Life?
Back in 1920, Latimer and Rodebush made an intriguing observation – a hydrogen atom in water is attracted not just to one atom but two. Fast forward to 1948, and Linus Pauling put another piece in the puzzle, emphasizing how these hydrogen atoms help form what’s known as a divalent bond, linking two atoms together.
So, how do we define a hydrogen bond today?
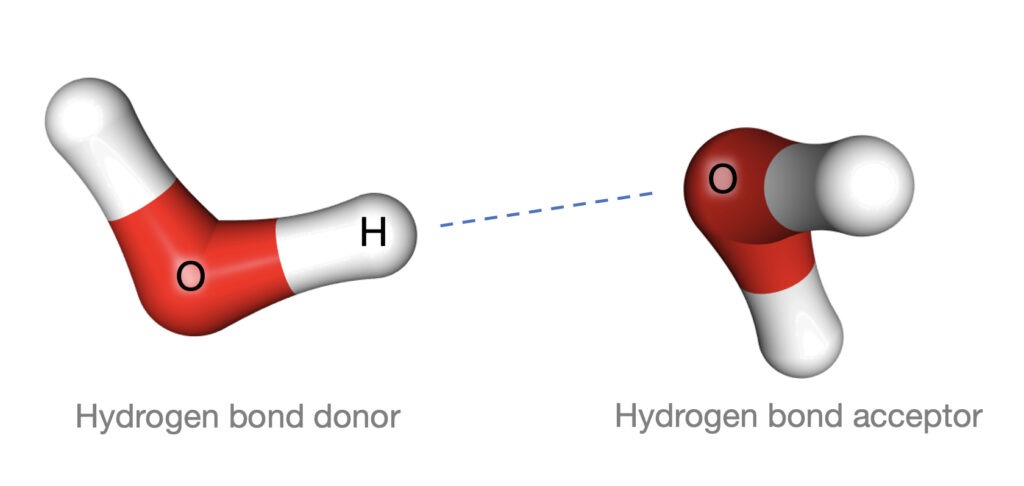
A hydrogen bond is primarily an electrostatic force of attraction. It occurs between a hydrogen atom, covalently bonded to a more electronegative donor atom or group, and another electronegative atom bearing a lone pair of electrons – the hydrogen bond acceptor.
In water, hydrogen bonds are a delicate interaction between the hydrogen and oxygen atoms within one water molecule, and the hydrogen’s attraction to a neighboring oxygen atom. Welcome to the H-O-H…OH2 hydrogen bond: a molecular noncovalent interaction that’s constantly forming and breaking.
In this light, water’s anomalous properties aren’t mere coincidences. They’re deeply connected to hydrogen bonding and water’s ability to form open tetrahedral hydrogen bond networks.
The cool thing is, hydrogen bonds are weaker than covalent bonds but stronger than just the random thermal jostling in water. This fine balance is what gives water its unique properties. Depending on the strength of these hydrogen bonds, water can expand to form a network or collapse into the gaps around other molecules.
Speaking of strength, in liquid water, the hydrogen bond energy is about 4-8 kcal/mol – no small amount, especially when compared to the average thermal collision fluctuation at 25°C, which is 5 times weaker. The strength of a water’s hydrogen bond is influenced by the bond angle and temperature. A straighter bond, closer to 180°, tends to be stronger. As the temperature goes up, the bonds weaken, affecting how water behaves.
Yes, water’s hydrogen bonds are pretty fleeting, with lifetimes ranging from 1 to 20 picoseconds. Broken bonds often reform quickly, maintaining the essential network of hydrogen bonds for water’s structure and function.
The subtle and dynamical properties of these hydrogen bond networks help explain why water behaves differently at different temperatures. For example, when solid, each water molecule is part of four hydrogen bonds arranged in a tetrahedral pattern. This structure is critical to ice formation and its anomalous lower density compared to liquid water.
So, it’s the hydrogen bonding that largely determines the peculiar and somewhat unusual properties of water that sustain life.
The beautiful thing is: the precise strength of the hydrogen bond seems almost tailor-made for life. What would happen if the strength of the hydrogen bond were just a tiny bit higher or lower?
Further reading: Strength of Hydrogen Bonds of Water Depends on Local Environment, 2012
More on From Atoms To Words:
▸ AI in Drug Discovery: Chasing Dreams, Facing Realities
▸ Large Language Models for Chemistry: Is the Beginning of a New Era?
▸ 60 Years in the Making: AlphaFold’s Historical Breakthrough in Protein Structure Prediction
Water’s Hydrogen Bonds: The Perfect Balance
So, let’s talk about the strength of water’s hydrogen bonds and why it’s such a big deal. Here’s the thing: even a small tweak in their strength can shake up water’s physical properties – we’re talking melting point, boiling point, density, and viscosity.
A little nudge can change the whole vibe.
It has been recognized that hydrogen bonds restrain protein molecules to their native configurations, and I believe that […] the significance of the hydrogen bond for physiology is greater than that of any other single structural feature.
Linus Pauling | 1931
If these bonds were stronger, water would behave more like a glass – more ordered, more structured. On the flip side, weakening these bonds would nudge water closer to the likes of methane and neon, which are all about feeble noncovalent interactions, like van der Waals.
This balancing act of hydrogen bond strength is also crucial in how water interacts with other substances. Stronger hydrogen bonds would increase water-water interactions, reducing its binding ability, while weaker bonds would reduce water-surface or water-solute interactions.
In terms of solubility, strong hydrogen bonding would aid in forming cavities that could accommodate small hydrophobic molecules, increasing their solubility.
So, what’s the takeaway here?
The strength of hydrogen bonds in water is kind of perfect. It’s not superglue strong, nor is it flimsy. It’s like a perfectly balanced zipper, gently holding things together while allowing just the right amount of flexibility.
More on From Atoms To Words:
▸ When Will RNA Structure Prediction Get Its AlphaFold Breakthrough?
▸ Multiscale Simulations of DNA: From Quantum Effects To Mesoscopic Processes
▸ Is Machine Learning Going to Replace Computational Chemists?
A Final Personal Touch
You know, when you really think about it – how everything in water just clicks into place, from its peculiar behavior at 4°C to the dynamics of its hydrogen bonds – it’s almost too easy to buy into the notion of a supernatural designer orchestrating it all. It’s the kind of cosmic fairy tale that eases our existential worries, a comforting thought in the vastness of the universe.
But let’s be real for a moment. Chalking it all up to intelligent design kind of feels like saying we won the cosmic jackpot just because the universe was in a giving mood. It’s a lovely narrative, sure, but it oversimplifies reality, its randomness, the sheer scale of probabilities.
The truth is, things just happen. Atoms find their sweet spots, bonds form, and molecules come to be, all following the relentless rules of quantum mechanics. Water’s hydrogen bonds are no exception.
Think of it like this: we’re living in a universe that’s just one among an infinite number of universes, each with its own set of starting conditions. It’s like a cosmic lottery, and our ticket just happened to have the winning numbers for life. Not because it was predestined, but because our universe rolled the dice just right.
Is this a satisfying answer? Probably not. The idea of a multiverse opens up more questions than it answers – do we live in a simulation? – leaving us in a bit of a pickle. But hey, whether it’s all a stroke of cosmic luck or there’s some supernatural designer at play, one thing’s for sure – water and its hydrogen bonds are downright amazing.
So, for now, let’s take a moment to marvel at the wonders of such a simple molecule: H2O.
If you enjoyed this dive into water’s hydrogen bonds and their relevance to Life, I’d love to hear your thoughts. Agree, disagree, or have a totally wild theory of your own? Let’s connect! Subscribe to my LinkedIn newsletter and let’s keep the conversation rolling.
EDIT on 12/02/24: I erroneously mentioned in the first paragraph that the density of water increases upon freezing. Thank you, Dr. Paul Stonaha, for spotting this ugly booboo.