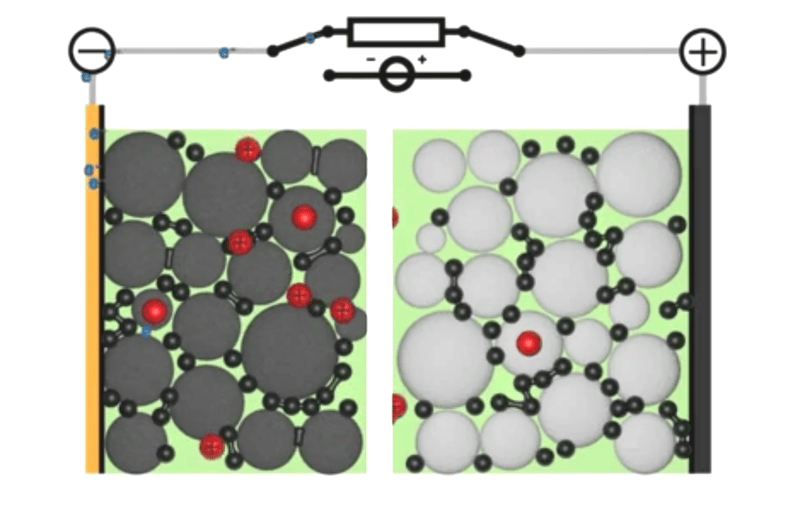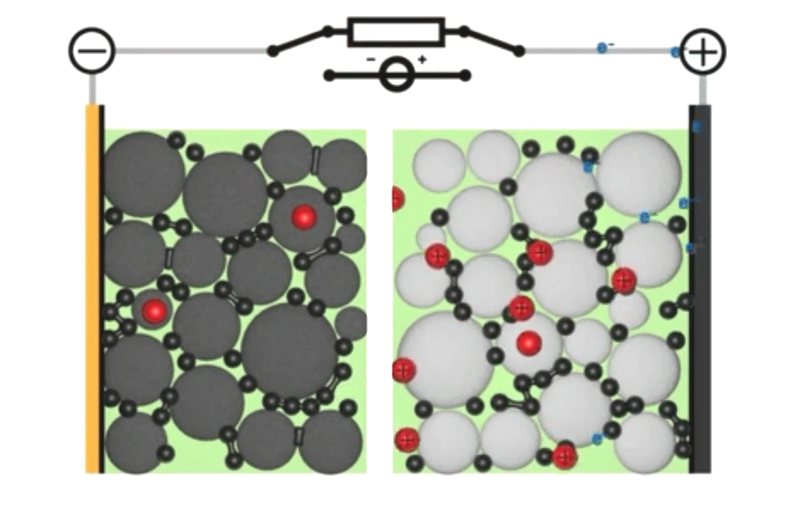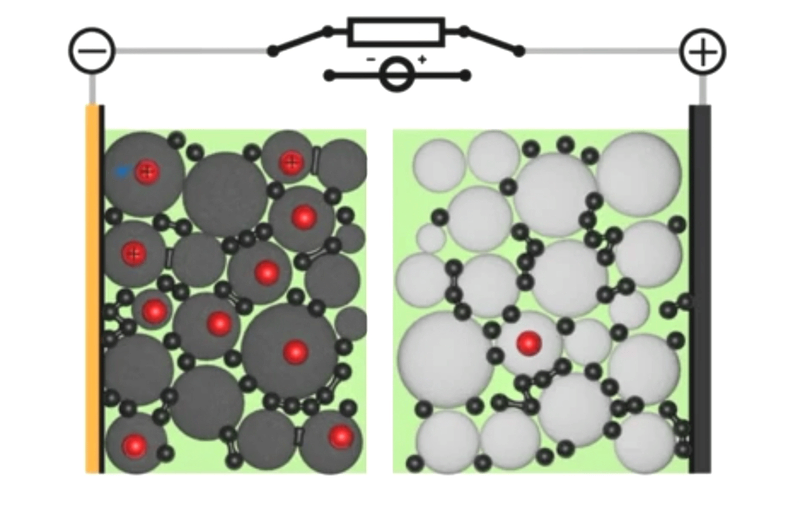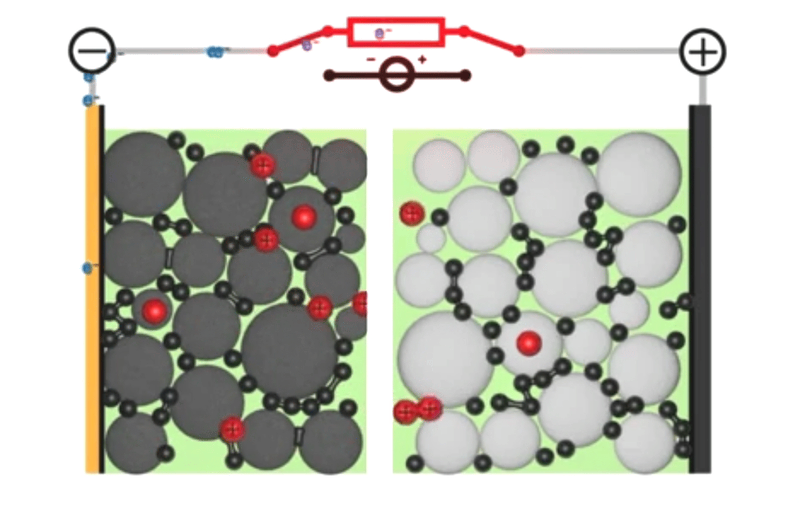
If you told my younger self, fresh out of a PhD in computational chemistry, that I’d end up working on simulations for next-generation battery design, he would have laughed at you. I mean, come on, I was all about DNAs and proteins – the little heroes that keep our bodies ticking against almighty entropy. But here’s the thing: life is full of surprises. And today, with my team at Quantistry, we’re collaborating with some of the world’s biggest players to push the boundaries of next-generation battery design and create a brighter, more sustainable future. So, what can atomistic simulations do for our electrical future? And why should R&D players bother learning about it? Well, my friends, let’s dive into the answers together.
Why should you care about Batteries?
When someone mentions batteries, you might conjure up images of bizarre pink bunnies bashing on their little snare drums. And you’re probably thinking, “What’s so fascinating about batteries anyway?”
In the search for renewable energy, batteries are an absolute game-changer.
The truth is, global climate change is an issue that’s getting more dire with each passing day. If we want to combat this problem and reduce our CO2 emissions, we have to find energy sources that don’t rely on carbon fossil fuels. Electrification is becoming vital in many areas (read transportation), which were previously dominated by oil or gas. However, long-term energy storage has always been a major challenge in the widespread implementation of clean technologies, like electric cars.
But that’s where lithium-Ion batteries come in. Unlike rechargeable batteries of the past, they have a much longer lifespan, higher efficiency, and are much safer to use. These advancements have led to the creation of a new era of portable devices, from smartphones to laptops and beyond.
It’s not a coincidence that in 2019, John Goodenough, Stanley Whittingham, and Akira Yoshino, were awarded the Nobel Prize in Chemistry for developing the “world’s most powerful battery.” Their innovation gives us the ability to effectively store and release sustainable energy when needed, delivering “the greatest benefit to humankind.”
So, it’s not just about those weird pink bunnies after all.
It’s about confronting the next formidable challenge of invention and reinvention. It’s about a global collective effort propelling us to new heights. It’s about the next-generation battery design that will shape our future.
Next-Generation Battery Design: A Combinatorial Minefield
Modern lithium-ion batteries, with their fancy rocking chair concept of intercalation and ion migration, are no joke, offering a specific energy of 170-220 Wh/kg. This is a significant improvement compared to previous technologies such as the lead acid battery, which only has an energy of about 30 Wh/kg.
But if we want to take this battery game to the next level and electrify the entire planet, we need to double that figure to at least 500 Wh/kg. It’s a lofty goal, but it’s the kind of ambition we need to create a cleaner and more efficient energy future.
And that’s why battery R&D is out there grinding away, exploring new chemistries and more sustainable, abundant raw materials. Lithium-metal batteries, for instance, are packing some serious heat with extremely high specific capacity. But let’s be real, their limited reserves make them unsustainable for long-term use. On the other hand, sodium-metal batteries are like the new kid on the block, with sodium being about 1000 times more abundant in the Earth’s crust than lithium.
Of course, nothing comes easy in the battery world.
Developing stable electrolytes and high-performance electrode materials is a major hurdle to overcome. Plus, let’s not forget about the safety concerns that arise with reactive metals like lithium and sodium.
These challenges all contribute to the sheer number of possible permutations for selecting active materials and electrolytes, which amounts to a mind-blowing 10100. That’s more than all the atoms in the entire universe. It’s like navigating a combinatorial minefield!
This means that the good old Edisonian trial-and-error approach, as Eng & Co. call it, just isn’t going to cut it anymore.
The solution? You guess right, my friend:
More on From Atoms To Words:
▸ Chemical Space to Material Discovery: Simulations and Machine Learning Leading the Way
▸ Quantum Chemistry of Molecule-Surface Adsorption: The 30-Year Struggle To Chemical Accuracy
Computer-aided Next-Generation Battery Design
Just as we’ve seen in previous stories about DNA simulations or metal-organic frameworks, computational techniques are transforming also battery research. Here are a few examples of how atomistic simulations, modelling, and machine learning, can support and accelerate next-generation battery design:
- High-throughput screening: DFT calculations can be used to screen large chemical spaces and predict properties of new battery materials. The most promising candidates can then be validated experimentally in a feedback loop.
- Deeper insight: Atomistic simulations can describe the structures and properties of less understood battery materials to provide insight into fundamental processes that are otherwise difficult to access. This includes ion diffusion mechanisms and electronic structure effects.
- Virtual experiments: Theoretical models and simulations can be used to conduct virtual experiments alongside to physical experiments, providing critical understanding of the battery system and observed phenomena.
- Validation: Even the most meticulously executed experiments can be subject to human error or cognitive bias in data interpretation. In this regard, computational approaches can help validate empirical data.
- Predictive capabilities: Models and simulations can predict the state of charge, state of health, and cycle life of batteries. When coupled with experimental measurements, this may enable a precise quantitative assessment of performance limits.
By successfully integrating simulations and experiments, we can establish a predictive framework for understanding the complex electrochemical processes occurring in batteries. With this deep knowledge, we can then engineer and control electrochemical reactions to build better batteries.
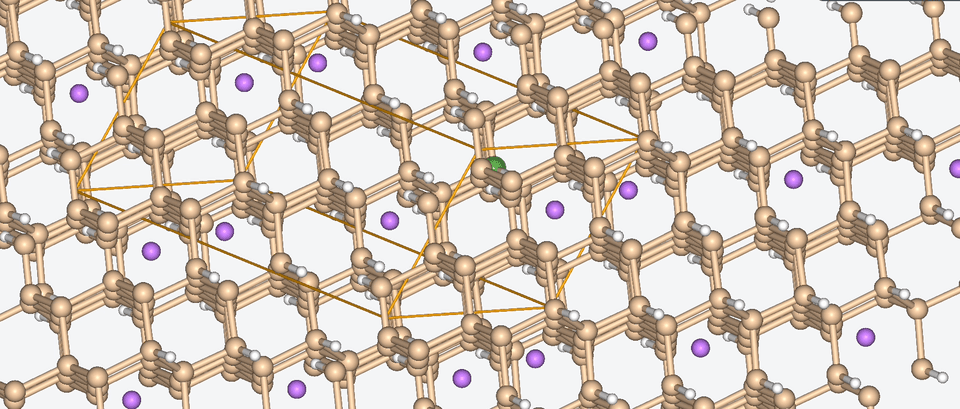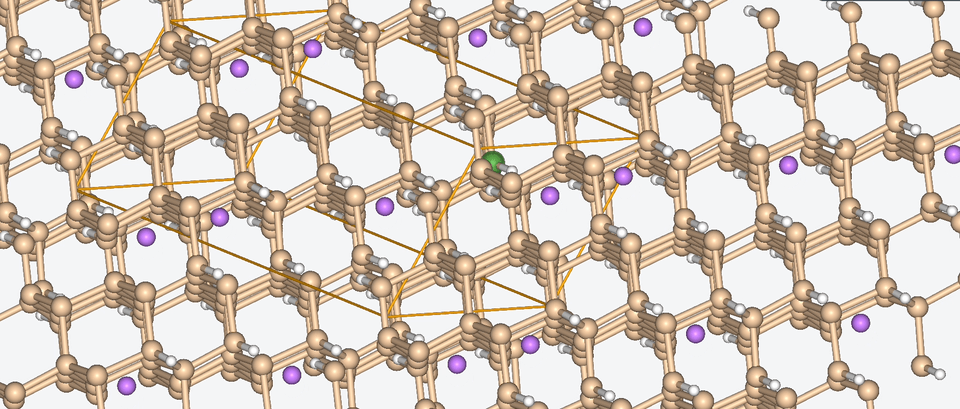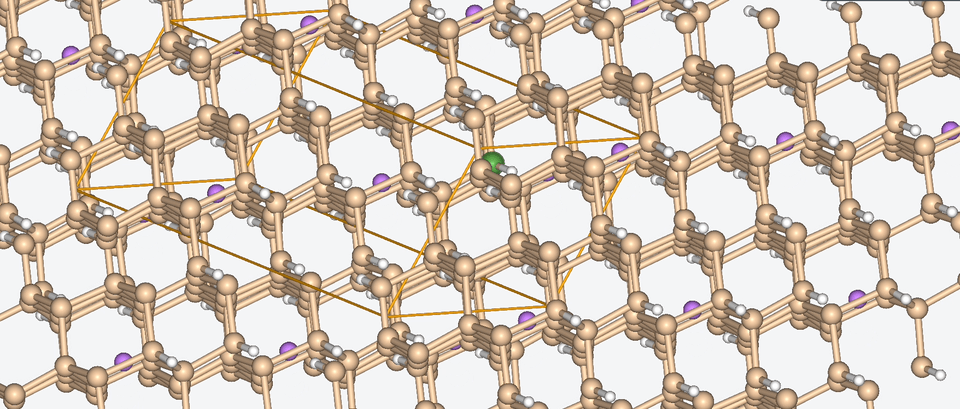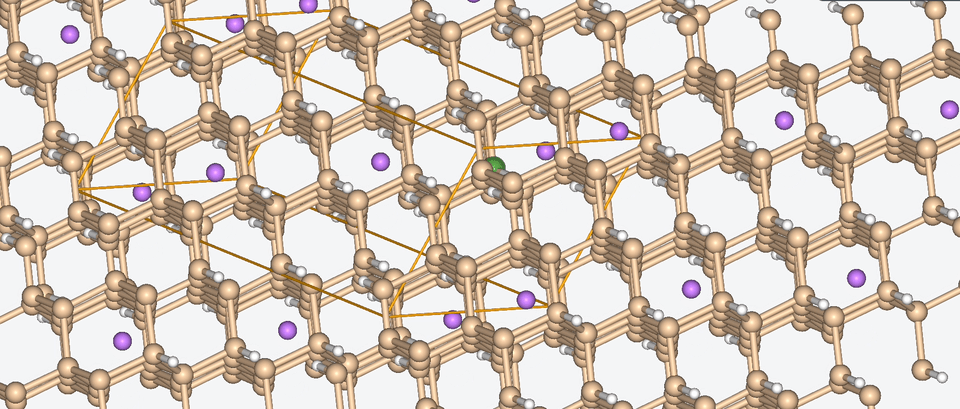
Let’s have a look at three case studies, in which computer-aided battery design was deployed.
More on From Atoms To Words:
▸ Computational Chemistry 2043: A Quantum Peep into the Future
▸ Large Language Models for Chemistry: Is the Beginning of a New Era?
Three success stories of computer-aided battery design
1. Exploring the chemical space of battery composition
In the magnificent world of battery composition, LiCoO2 is the dear cathode material that possesses exceptional properties. However, its exorbitant cost and the dark cloud of unethical cobalt mining have triggered a frantic quest for alternatives.
In a transformative DFT study dating back to 1998, Ceder & Co. sought to explore the chemical space by meticulously swapping non-transition metals with their transition metal counterparts.
They hit the jackpot when they replaced cobalt with aluminum. They then went and proved their theoretical predictions with experiments. The results of X-ray diffraction and scanning transmission electron microscopy confirmed that the new cathode material exhibited reversible charge/discharge behavior for around 10 cycles.
Since then, the combo of theory and experimentation has catapulted the field of battery research.
Now, with better computational power and experimental techniques, we’ve been making leaps and bounds in the development of sustainable, high-performance batteries that can keep up with our increasingly electrified world.
2. Unveiling the Key Principles of Redox Processes
One of the greatest discoveries reached by a combination of theory and experiment is of the simultaneous cationic and anionic redox reactions in lithium-excess layered oxide cathode materials.
Traditionally, the reversible charging/discharging process is attributed to the cathode’s cations. However, lithium-excess systems, adorned with precious metals like ruthenium or iridium, introduces a fascinating twist. Here, the oxide anions themselves can partake in the exhilarating dance of oxidation and reduction, granting access to vastly superior theoretical capacities.
But there’s a catch. These precious metals bear their title for a reason—they come at a steep price.
In light of this, the diligent minds of battery R&D embarked on a quest for alternative compounds that could harbor the same electrifying potential. They introduced Li5FeO4 cathodes, a glimmer of hope amidst the scarcity of resources.
Now, this is where Zhan & Co. make their grand entrance.
Their DFT calculations identified the lithium-excess Li6-O configuration as a key player in enabling the reversible O−/O2− redox behavior. Now, I won’t get into the technical nitty-gritty of it all, but basically, they performed density of states calculations to find that O− in the Li6-O configuration represents labile oxygen states, which can then undergo reversible transformation to O2−. This forms the basis of anionic redox in Li5FeO4 cathodes. Zhan & Co. then performed experiments, like ex situ Raman spectroscopy, x-ray diffraction, and x-ray absorption spectroscopy, to make sure their predictions were on point.
So, the bottom line is that Zhan & Co.’s research is a big deal. They’ve given us a framework for understanding redox processes and some guiding principles for designing improved lithium-ion battery systems.
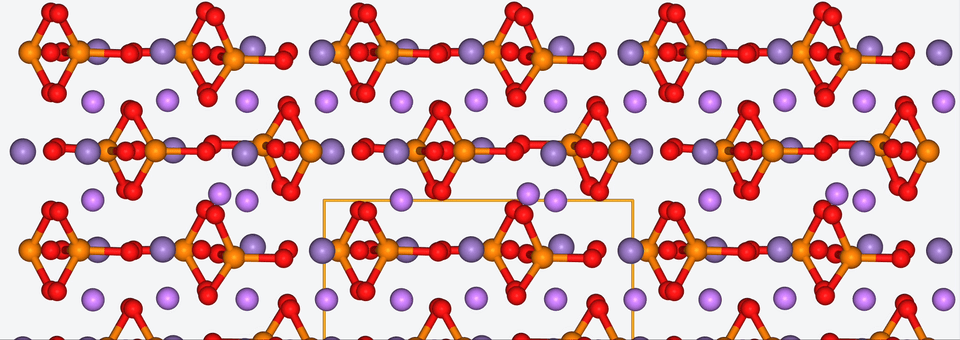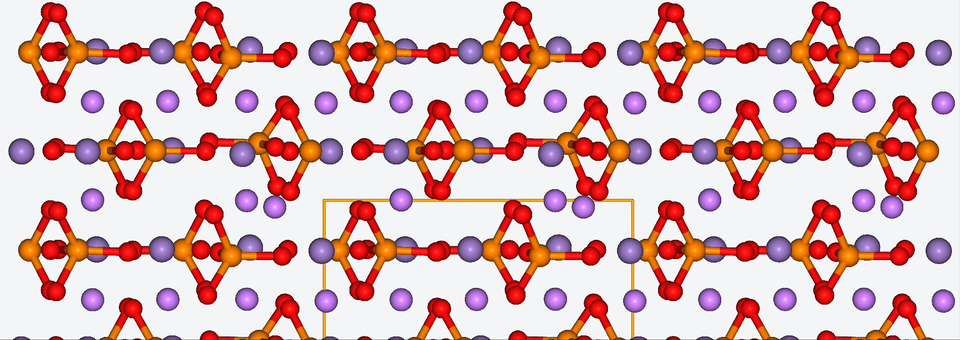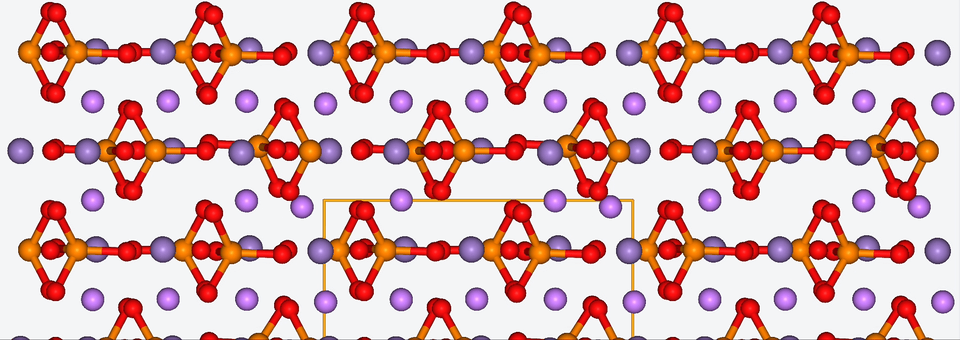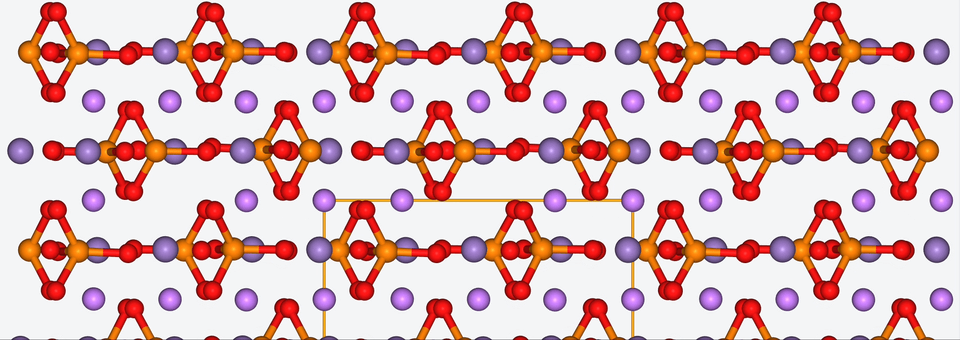
3. Zooming into the atomistic behavior of SEI films
When it comes to high-performance lithium-ion batteries, the solid electrolyte interphase (SEI) film is like the bouncer at a club. It stands guard at the entrance of the anode interface, making sure nothing sketchy gets in.
The SEI film is made of decomposed electrolyte moieties and acts as a barrier to prevent further electrolyte decomposition and maintain the electrochemical stability of the electrolyte. But despite its critical role, there’s still a lot we don’t know about this enigmatic component.
To shine some atomistic light on the SEI film, Baba & Co. employed ab initio molecular dynamics, a method with which we are well-acquainted, to analyze the structural, electronic, and free energy properties of Li ion migration across the interface between the anode and the SEI film.
Drawing from their data, Baba & Co. created a map of the electric landscape that Li ions have to navigate when moving between the anode and cathode, providing an atomistic level understanding of Li-ion transport at battery interfaces.
With this knowledge, we can work towards developing more efficient and sustainable high-performance batteries in the future.
More on From Atoms To Words:
▸ Modeling the Origins of Life: Quantum Simulations of the Primordial Soup
▸ Multiscale Simulations of DNA: From Quantum Effects To Mesoscopic Processes
▸ All-Atom Molecular Dynamics of SARS-CoV-2: The Computational Microscope’s View of 305 Million Atoms
A final personal touch
Batteries are some seriously complex systems. I hear you: this stuff is hard. We’re talking multicomponent, dynamical, and jam-packed with chemical-physical phenomena that occur during charge and discharge.
If you want to dig deeper, check out this Science review by Eng & Co. – It’s a solid starting point.
This article aligns with what I have been learning through months of work at Quantistry. It all comes down to a simulation-experiment feedback loop, where we use theory to guide our experimental design, and then take those experimental results back to the drawing board to refine our simulations.
And here’s where things get really exciting. With the rise of machine learning, high-throughput quantum chemistry methods, and quantum computing, we’re poised to make some serious breakthroughs in battery materials design and discovery.
So, let’s get after it. By teaming up and blending the power of theory and experiment, we can create a more secure energy future for all of us.
If you enjoyed this dive into battery materials and their marvelous features, I’d love to hear your thoughts. Agree, disagree, or have a totally wild theory of your own? Let’s connect! Subscribe to my LinkedIn newsletter and let’s keep the conversation rolling.